(by A. Schaafsma MD PhD)
After taking over a practice in clinical neurophysiology at the Martini Hospital Groningen from my predecessor Rud Brenninkmeijer in 1998, I was confronted with a peculiar type of apparatus that I had not encountered before: the oculoplethysmograph. After instilling a drop of local anaesthetic in both eyes, cups were placed over the sclera allowing the apparatus to exert negative pressure to the patient's eyes, temporarily blocking arterial inflow. When pressure was gradually released arterial pulsation returned and could be recorded for both sides independently, thereby providing information on the arterial blood pressure wave in each central retinal artery. Pulsation returned at lower pressures in the eye ipsilateral to carotid artery stenosis depending on the amount of collateral flow.

In the era of HIV, hospital epidemiologists rapidly concluded this apparatus was no longer to be used, since the eye cups could not be properly disinfected. In search for an alternative way to assess intracranial hemodynamics we (technicians and physicians of our department) turned to transcranial Doppler (TCD). At first we recorded middle cerebral artery blood velocity (MCAV) few hours before and on days 0-3 after carotid endarterectomy aiming to detect postoperative hyperperfusion syndrome as a complication potentially leading to severe morbidity or mortality.

Sitting on the floor with the recordings of approximately 40 patients gathered around me I was struck by the great variability of our findings: sometimes the MCAV increased dramatically after surgery but in some patients it could also decrease. Soon I realized we needed to be informed about arterial blood pressure as well, under the assumption that this is the driving force behind middle cerebral artery flow velocity.
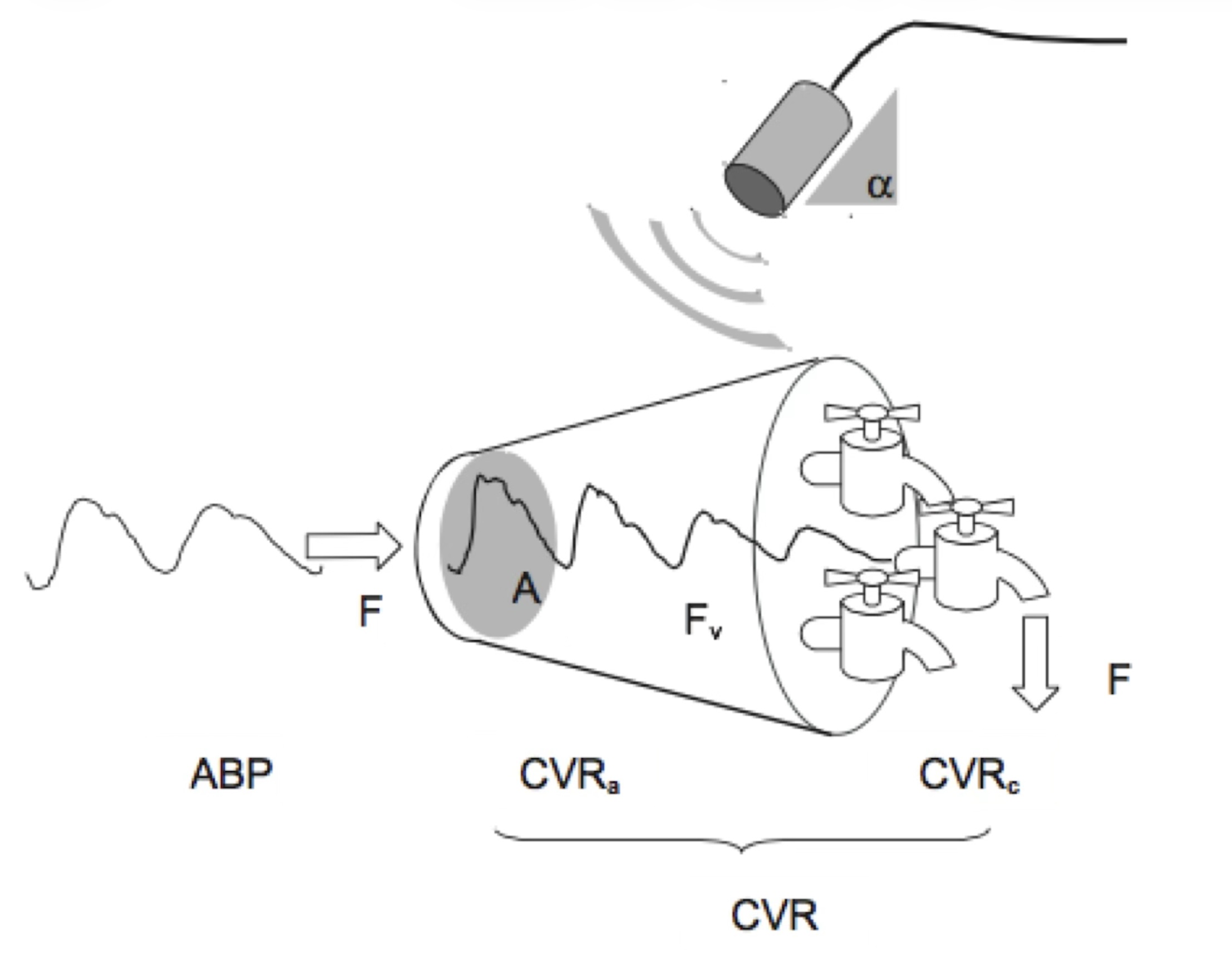
I was looking for a better understanding how the MCAFV signal related to the different physiological parameters, such as heart rate, end-tidal CO2 and arterial blood pressure. I realized that the MCAFV is a conducting artery positioned between the large feeding arteries and the outflow arterioles and cerebral capillary networks of the larger part of a cerebral hemisphere.

From a colleague, Klaas Hoogenberg, we borrowed a non-invasive blood pressure (NIBP) measuring device, the Ohmeda Finopress 2300. This apparatus allowed us to make a continuous recording of arterial blood pressure by placing a cuff around a finger. A servo mechanism maintained a continuous volume in the finger (volume clamp), requiring rapid changes in inflation pressure. The pressure required mirrored the pressure exerted by the arterial pulse. This led to detailed and synchronous information on wave morphology of both MCAV and NIBP. Combining the two signals seemed promising so these measurement techniques were used on patients with carotid stenosis and on patients with autonomic dysfunction (2000).

There was remarkably little agreement between both signals. What corresponded best was the heart rate and thus the duration of a single heart cycle. We defined the pulse flow velocity mismatch (PFVM) and later the pulsatile apparent resistance (PaR) as indices expressing the (dis)agreement between both signals. In 2004, the PaR was patented as a blood pressure corrected pulsatility index, since it expressed the difference between the apparent resistance (velocity/pressure) during end diastole and the 2nd systolic peak divided by the average apparent resistance. For quite some time we focused on the second peak in both signals since this correlated best with heart rate and CO2-reactivity.

Based on an EU and US patent on PaR technology, Neuromon B.V. successfully applied for a IAG-grant allowing the company to buy three set-ups combining a Rimed TCD, a next generation Finometer and a capnograph mounted on a single equipment rack. Singals were processed by an Apple mini-mac running customized LabVIEW software. A collaboration agreement was established with the University Hospital Groningen, the Erasmus Hospital Rotterdam and the Martini Hospital Groningen. For some time data was collected in carotid artery patients and patients with traumatic brain injury (2007).

Around this time (2009) I analyzed MCAFV signals during deep breathing by calculating bin averages based upon the RR'-interval in the ECG. It became clear that there are two peaks in systole, both in the NIBP as well as MCAV signal. I was able to show that there is a considerable difference in variance between the Sys1 and Sys2 components, suggesting they have a different physiological origin. From then on, I started to focus on the Sys1 component more. The question arose why the Sys1 was less dependent on variation in RR'-interval and thus cardiac stroke volume than the Sys2.
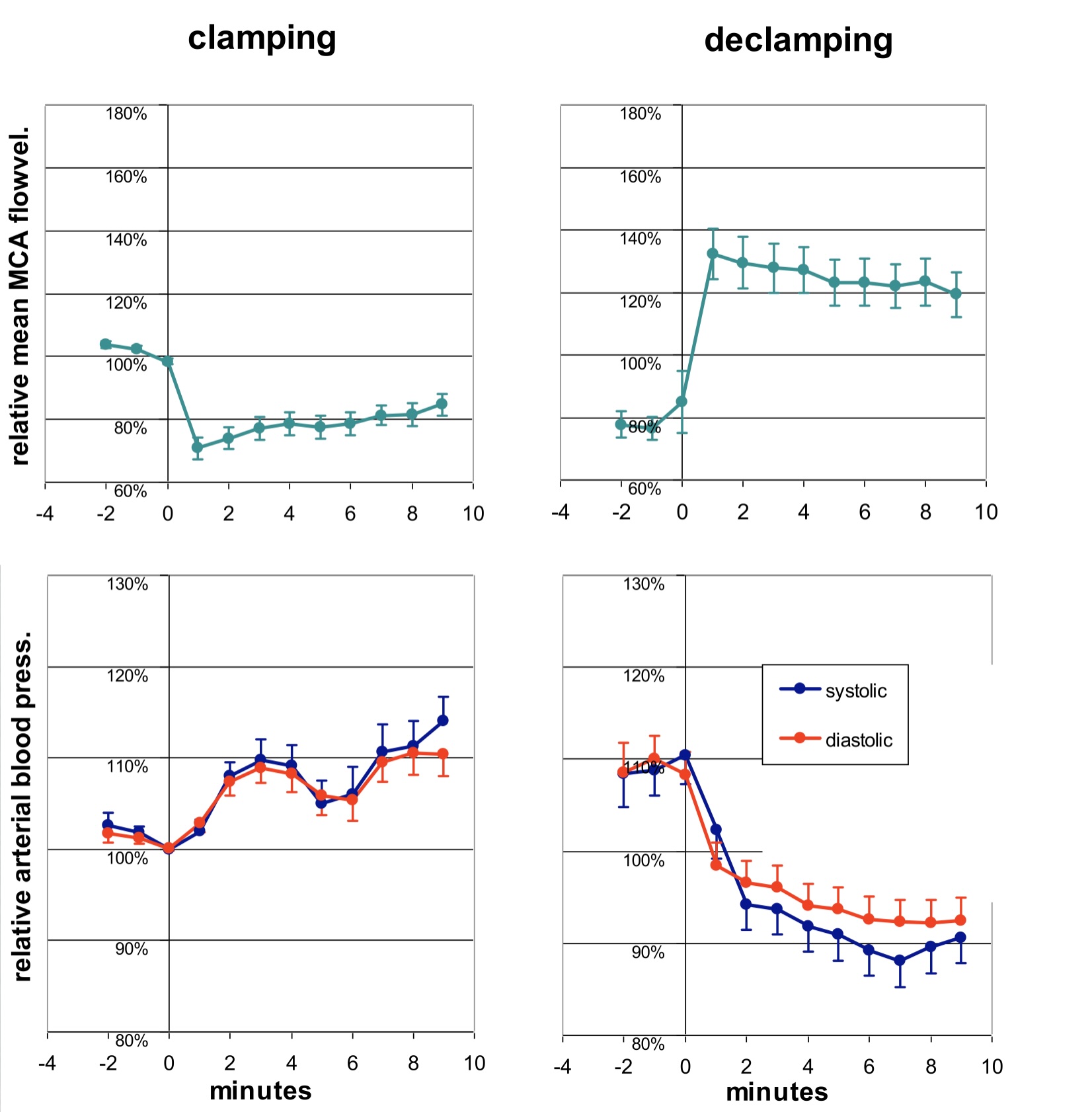
During carotid endarterectomy many hours of simultaneously recordings of ECG, bilateral MCAFV and ABP were obtained. I was able to document changes during clamping and declamping. Also, I was able to study changes in the two signals in response to the administration of specific drugs, such as norepinephrine, dopamine and atropin. However, application of PaR-technology in patients with traumatic brain injury (TBI) was cumbersome. Prolonged recordings were difficult to obtain since these required a fixation band mounted on the patients head. This made me return to the MCAFV signal itself, settling with intermittent monitoring: repetitive recordings of a few minutes with a hand held probe.

In 2012 new parameters were proposed to better describe the pulsatile signal: Sys1, Sys2 and D560 (the diastolic blood velocity 560 ms after upbeat onset). It was soon realized that these parameters were also applicable to the ABP signal. Together with Annika de Goede software was developed for online and automated detection of these parameters allowing their realtime presentation on screen. Nowadays this is an important add-on during autonomic function testing at our lab.

In 2013 a collaboration agreement was established with Compumedics DWL allowing Neuromon to market the software for automated parameter detection. A dynamic library was built and offered exclusively to end users of DWL TCD equipment with QL software.

For the MCAFV we provided normative data obtained in over 100 Dutch volunteers ranging from 18 to 80 years old. This allowed the calculation of so-called Z-scores, describing how many standard deviations a given measurement was above or below the value expected for that age. Combining the Z-scores for Sys1, Sys2, D560 and HR (heart rate) in a single radar plot enables a more rapid interpretation of the MCAFV signal. It can also be used for follow up measurements after CEA and for monitoring changes in intracranial hemodynamics during autonomic function testing.

In 2014 I published the theory of arterial acceleration in Medical Hypotheses. This paper explains the biphasic appearance of the systolic peak by proposing a shortlasting contraction in the smooth muscle layers of conducting arteries during Sys1 that adds to the ejection phase of the heart indicated by Sys2.
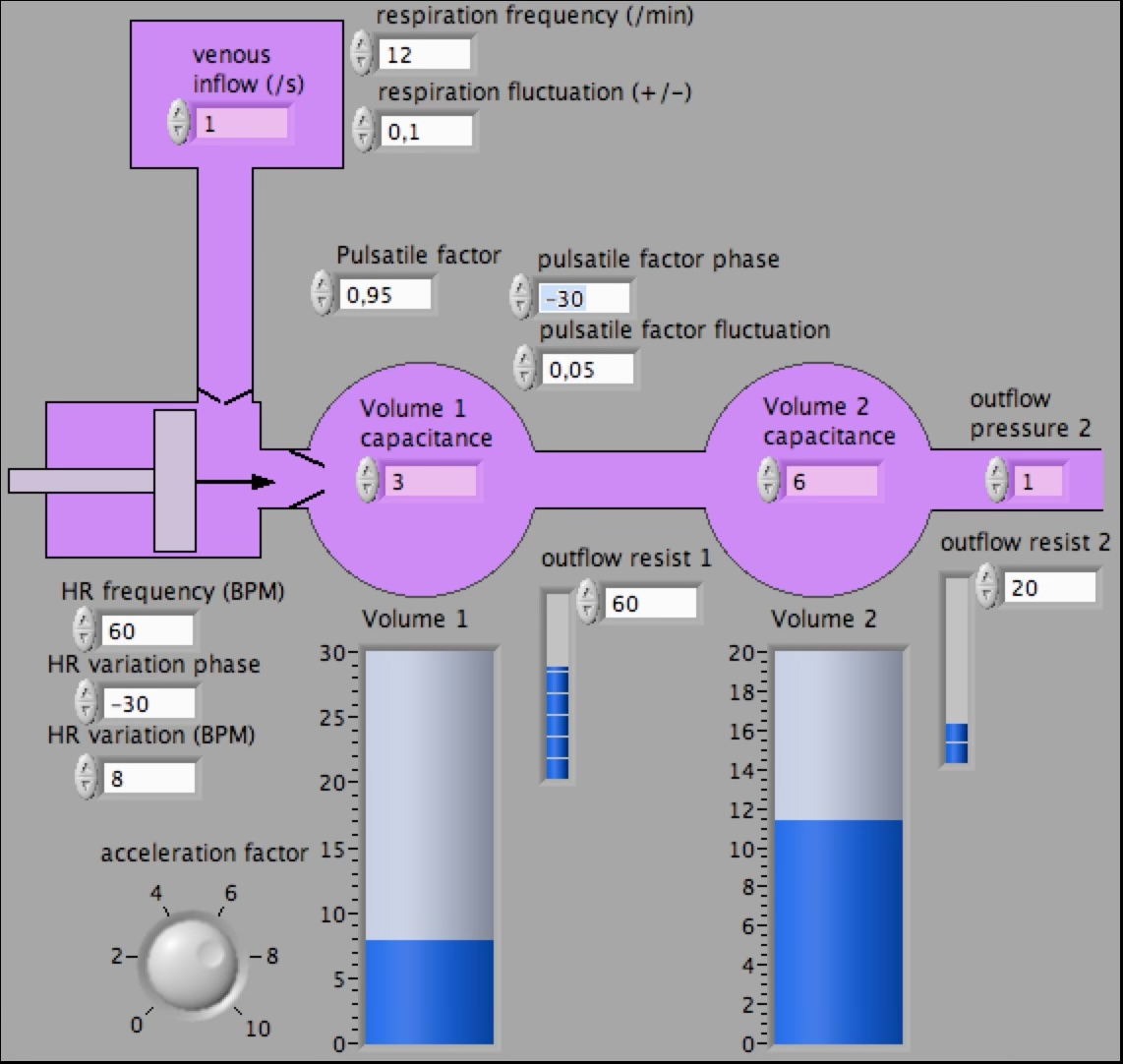
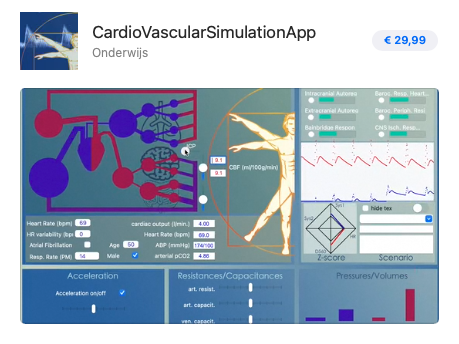
To explain how arterial acceleration enhances the penetration of the pressure wave into all the bodies capillary systems a computer model was designed evolving from a simple LabView based model to the CardioVascularSimulationApp that can be downloaded from the AppStore.

In 2017 we described changes in waveform characteristics during repetitive fluid challenges in patients with sepsis. We described the newly defined parameters for the follow up in patients undergoing carotid endarterectomy. A study was undertaken showing differences in waveform during standing and sitting. Characteristic changes were described for MCAFV and NIBP during hyperventilation, normoventilation and CO2-retention. S1-PaR was shown to correlate with intracranial pressure elevation (2025).

The theory of arterial acceleration obtained little attention until in 2025 evidence was found in a group of patients with aortic stenosis showing that from proximal to distal a phase of laminar flow (Sys1) increasingly replaces the turbulent phase of cardiac outflow (Sys2). The Sys1 is an addition to cardiac ejection and represents energy added by the conducting arteries to the pressure wave from the heart.

It is Neuromon's mission to increase the awareness of the theory of arterial acceleration and to implement the new parameters for the description of pulsatile signals in medical systems. In the end these goals are expected to support medical decision making. In 2026 the new parameters are commercially available in the TCD system of Compumedics DWL Germany and, with slight adaptations, in a blood pressure device for the outpatient clinic manufactured by Uscom Australia.
